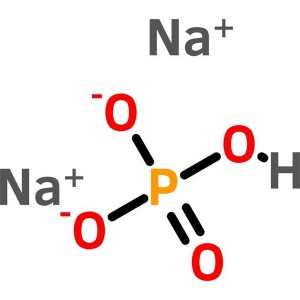
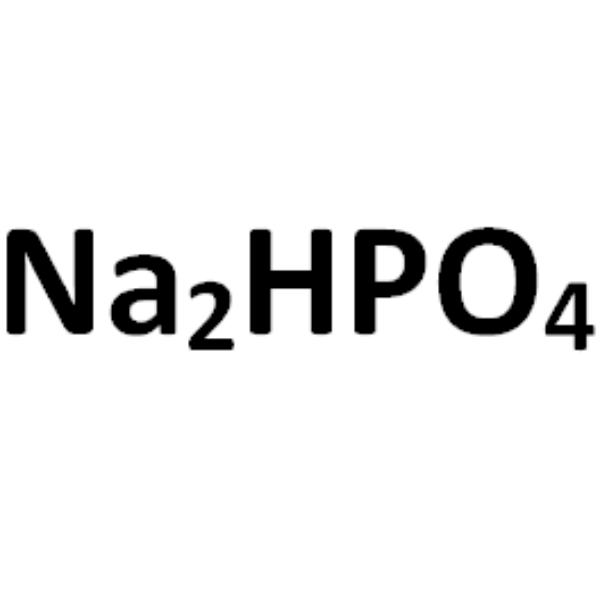Sodium Phosphate Dibasic CAS 7558-79-4 Purity >99.5% (Titration) Biological Buffer Ultra Pure
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer and supplier of Sodium Phosphate Dibasic (CAS: 7558-79-4) with high quality, commercial production. Ruifu can provide worldwide delivery, competitive price, excellent service, small and bulk quantities available. Welcome to order. Please contact: alvin@ruifuchem.com
| Chemical Name | Sodium Phosphate Dibasic |
| Synonyms | DSP; Sodium Phosphate Dibasic Anhydrous; Disodium Phosphate; Dibasic Sodium Phosphate; Disodium Hydrogen Phosphate; Disodium Hydrogen Phosphate Anhydrous; di-Sodium Hydrogen Phosphate Anhydrous; Sodium hydrogenphosphate |
| CAS Number | 7558-79-4 |
| Stock Status | In Stock, Production Scale Up to Tons |
| Molecular Formula | Na2HPO4 |
| Molecular Weight | 141.96 |
| Melting Point | 243.0~245.0℃ |
| Density | 1.064 g/mL at 20℃ |
| Stability | Stable. Incompatible with Strong Acids. Hygroscopic. |
| Sensitive | Hygroscopic |
| COA & MSDS | Available |
| Origin | Shanghai, China |
| Brand | Ruifu Chemical |
| Item | Specifications |
| Appearance | White Powder |
| Purity / Analysis Method | >99.5% (Titration, on Dried Basis) |
| Loss on Drying | <1.00% (120℃, 4h) |
| Water Insoluble Matter | <0.20% |
| pH (5% Solution at 25℃) | 8.7~9.3 |
| Solubility (5%, H2O) | Colorless Clear Solution |
| Arsenic (As) | <3ppm |
| Heave Metals (as Pb) | <10ppm |
| Iron (Fe) | <5ppm |
| Chloride ( Cl) | <10ppm |
| Sulfate ( SO4) | <50ppm |
| Potassium ( K) | <50ppm |
| Calcium (Ca) | <10ppm |
| Copper (Cu) | <5ppm |
| Total Metallic Impurities | <200ppm |
| UV Absorbance/280 nm | <0.05 (1 M, Water) |
| ICP | Confirms Na and P Components |
| X-Ray Diffraction | Conforms to Structure |
| Test Standard | Enterprise Standard; FCC Standard |
| Usage | Biological Buffers and etc. |
Package: Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement.
Storage Condition: Keep the container tightly closed and store in a cool, dry and well-ventilated warehouse away from incompatible substances. Protect from light and moisture. Keep away from fire and heat source. Shall not be co-stored with moist or toxic substances. Avoid rain and sun exposure during transport. When loading and unloading should be light to prevent packaging damage. When there is a fire, it can be suppressed by using water, sand and fire extinguisher.
Shipping: Deliver to worldwide by air, by sea, by FedEx / DHL Express. Provide fast and reliable delivery.

Hazard Symbols Xi - Irritant
Risk Codes
36/37/38 - Irritating to eyes, respiratory system and skin.
Safety Description
S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
S36 - Wear suitable protective clothing.
S24/25 - Avoid contact with skin and eyes.
UN IDs UN 3077 9 / PGIII
WGK Germany 3
RTECS WC4500000
FLUKA BRAND F CODES 3
TSCA Yes
HS Code 2835252000
Sodium Phosphate Dibasic (CAS: 7558-79-4), Usage: 1) buffering 2) improving agent 3) emulsifier 4) nutrition supplement 5) antioxidant additive. Sodium Phosphate Dibasic is highly hygroscopic and water soluble. Useful in conjunction with Sodium Phosphate, Monobasic in the preparation of biological buffers. It is commonly used in biological assay buffers. Sodium Phosphate Dibasic is reagent with high buffering capacity for molecular biology, biochemistry, and chromatography. Sodium Phosphate Dibasic is used in food and water treatment in conjunction with trisodium phosphate. In foods, it is used to adjust pH. In water treatment, It retards calcium scale formation. It is also found in some detergents and cleaning agents. This product is known to be used in many applications including the purification of antibodies.
Sodium Phosphate Dibasic is used to adjust pH of liquid. Disodium phosphate can be used as an excipient, such as buffer, chelating agent. Pharmaceutical excipients, or pharmaceutical auxiliaries, refer to other chemical substances used in the pharmaceutical process other than pharmaceutical ingredients. Pharmaceutical excipients generally refer to inactive ingredients in pharmaceutical preparations, which can improve the stability, solubility and processability of pharmaceutical preparations. Pharmaceutical excipients also affect the absorption, distribution, metabolism, and elimination (ADME) processes of co-administered drugs.
-

Sodium Phosphate Dibasic CAS 7558-79-4 Purity >...
-
Disodium Succinate Hexahydrate CAS 6106-21-4 Pu...
-
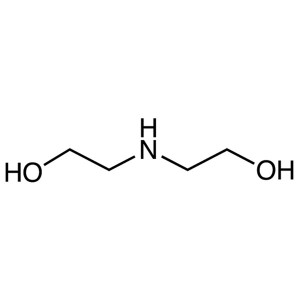
Diethanolamine (DEA) CAS 111-42-2 Purity >99.5%...
-
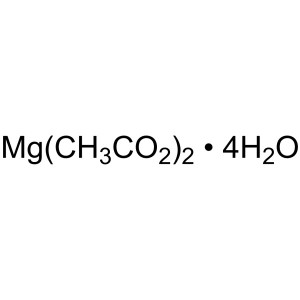
Magnesium Acetate Tetrahydrate CAS 16674-78-5 P...
-
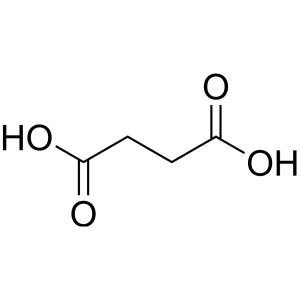
Succinic Acid CAS 110-15-6 Purity >99.5% (Titra...
-
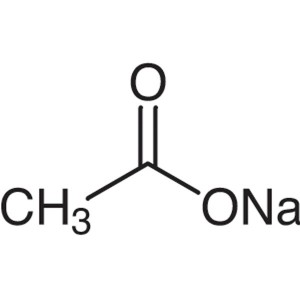
Sodium Acetate CAS 127-09-3 Purity >99.5% (Titr...
-
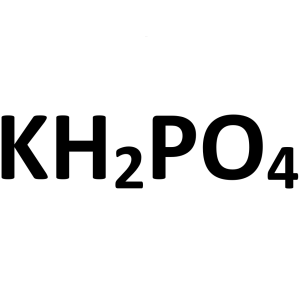
Potassium Phosphate Monobasic CAS 7778-77-0 Pur...
-
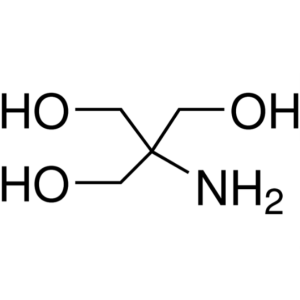
Tris Base CAS 77-86-1 Purity ≥99.50% (Titration...
-
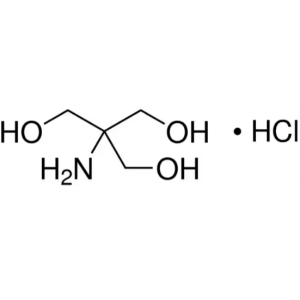
Tris-HCl CAS 1185-53-1 Purity 99.50%~101.0% (Ti...
-
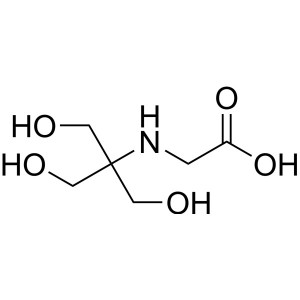
Tricine CAS 5704-04-1 Purity>99.5% (T) Biologic...
-
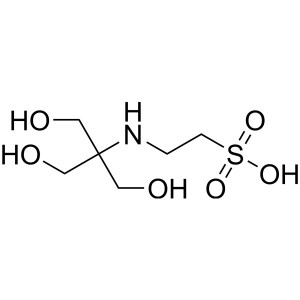
TES CAS 7365-44-8 Purity >99.5% (Titration) Bio...
-
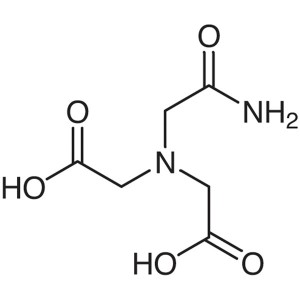
ADA CAS 26239-55-4 Purity >99.0% (Titration) Bi...
-
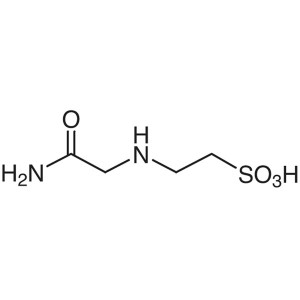
ACES CAS 7365-82-4 Purity>99.0% (Titration) Bio...
-
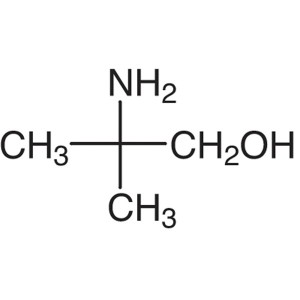
AMP CAS 124-68-5 Purity >99.0% (GC) Biological ...
-
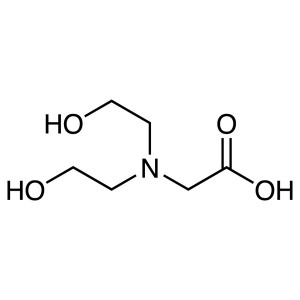
Bicine CAS 150-25-4 Purity >99.5% (Titration) B...
-
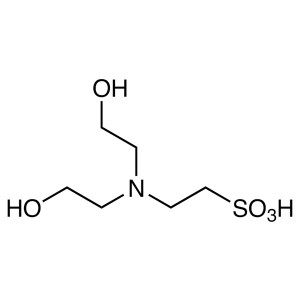
BES CAS 10191-18-1 Purity >99.5% (Titration) Bi...